Achondroplasia & FGFR3
This page was produced as an assignment for Genetics 677 at UW-Madison, Spring 2010
Discussion
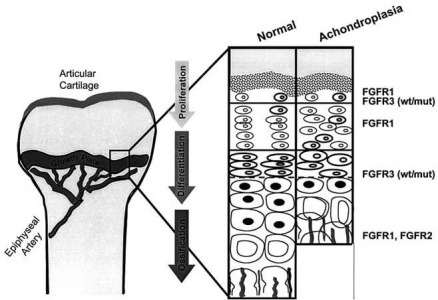
The function of FGFR3 in cell signaling, and specifically, bone ossification seems relatively well understood. FGFR3 serves to repress the number of chondrocytes that proliferate and mature into bone at the growth plate. When the mutation for achondroplasia occurs in the transmembrane region of the receptor, there is a gain-of-function and ossification is repressed even more. Because of this, a possible treatment option would be to interfere with binding at the receptor. This would reduce its activity, causing an increase in bone ossification, and hopefully more bone growth in those with achondroplasia.
As can be seen in the String protein interaction network FGFR3 shares many of the same substrates as the other receptors in the FGFR family. However, it has one domain in the the extracellular region that is different from the other FGFR receptors, it has an IG domain and two IGc2 domains, compared to the others that just have three IGc2 domains. This distinguishing feature, even if it is slight, could be a good way to specifically target FGFR3 for drug therapies for achondroplasia.
The one bit of information that seems to be yet unknown about FGFR3 in bone ossification is specifically which FGF is involved. I would propose that this is the next step in researching achondroplasia.
Proposed Research
In order to determine which fibroblast growth factor binds FGFR3 during bone ossification I would use a mouse as a model organism. Mice have been successfully used in a number of other studies involving the achondroplasia mutation in FGFR3, and have a mutant phenotype very similar to that of humans. [1] fhere are as many as 23 FGFs in the body, so to narrow down the possibilities I would first use a microarray to determine which FGFs are expressed in the bone growth plate tissue, and then focus on those.
Next, I would create knockout mice for those FGFs. While, FGFs can be crucial for a number of pathways within the body, knockouts for different FGFs have been successful in other experiments. [1] Additionally, the mouse would not have to live for long, as the mutant phenotype can be observed in newborn mice.
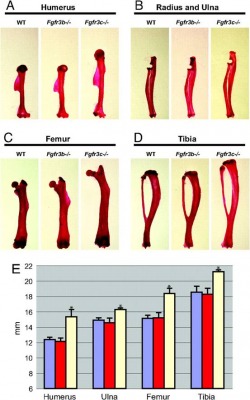
Since I will, hopefully, be removing the FGF binds to FGFR3 and causes the repression of ossification, this should actually result in a phenotype that shows more ossification, and has longer bones. It should be very similar to a phenotype observed in a study in which they created FGFR3 knockouts. The FGF that produces this result should be the one involved in bone ossification.
Next, I would create knockout mice for those FGFs. While, FGFs can be crucial for a number of pathways within the body, knockouts for different FGFs have been successful in other experiments. [1] Additionally, the mouse would not have to live for long, as the mutant phenotype can be observed in newborn mice.
Since I will, hopefully, be removing the FGF binds to FGFR3 and causes the repression of ossification, this should actually result in a phenotype that shows more ossification, and has longer bones. It should be very similar to a phenotype observed in a study in which they created FGFR3 knockouts. The FGF that produces this result should be the one involved in bone ossification.
Knowing the specific FGF that is involved in ossification can be useful in a number of ways. We would know the ligand structure that is preferred in this process, which could allow us to create a similarly structured compound that could interfere with the binding by blocking up the receptor, thus reducing the repression of ossification. Otherwise, compounds that bind to that specific ligand and prevent it from binding with the receptor could be researched as well.
Future Directions
Beyond my proposed research project, other directions could be taken to learn more about FGFR3 and possible drug targets.
For example, a compound called heparan sulfate is necessary in the binding of FGF to the receptor. This compounds role can be seen in the signaling pathway. Analyzing it with a structural-based chemical genetics program could yield other compounds that could interfere with binding at the receptor.
Additionally, chemical genetics screens could be used to determine if any other compounds can interfere with receptor binding. This video illustrates how a chemical genetics screen can be set up to test a large quantity of compounds at once, and observe for changes in a cell that would indicate an interaction with a specific small molecule. Video from the 2002 Lecture "Scanning Life's Matrix: "Genes, Proteins, and Small Molecules."
For example, a compound called heparan sulfate is necessary in the binding of FGF to the receptor. This compounds role can be seen in the signaling pathway. Analyzing it with a structural-based chemical genetics program could yield other compounds that could interfere with binding at the receptor.
Additionally, chemical genetics screens could be used to determine if any other compounds can interfere with receptor binding. This video illustrates how a chemical genetics screen can be set up to test a large quantity of compounds at once, and observe for changes in a cell that would indicate an interaction with a specific small molecule. Video from the 2002 Lecture "Scanning Life's Matrix: "Genes, Proteins, and Small Molecules."
References
[1] Aviezer, Golembo, and Yayon. Fibroblast Growth Factor Receptor-3 as a Therapeutic Target for Achondroplasia- Genetic Short Limbed Dwarfism. Current Drug Targets. 2003. 4, 353-365
[2] Eswarakumar and Schlessinger. Skeletal overgrowth is mediated by deficiency of a specific isoform of fibroblast growth factor 3. National Academy of Sciences of the USA. 2007. http://www.pnas.org/content/104/10/3937.full
[1] Aviezer, Golembo, and Yayon. Fibroblast Growth Factor Receptor-3 as a Therapeutic Target for Achondroplasia- Genetic Short Limbed Dwarfism. Current Drug Targets. 2003. 4, 353-365
[2] Eswarakumar and Schlessinger. Skeletal overgrowth is mediated by deficiency of a specific isoform of fibroblast growth factor 3. National Academy of Sciences of the USA. 2007. http://www.pnas.org/content/104/10/3937.full